Electrophoresis
Migration of Ions in an applied Electric Field.
The first recorded measurements of electrophoretic phenomenon were performed in 1861 by QUincke.
Tiselius. by describing his moving boundary apparatus in 1937, was instrumental in popularizing the utility of electrophoresis to the biochemist.
Electrophoresis is the migration of charged particles or molecules in a medium under the influence of an applied electric field.
Many important biological molecules, such as amino acids, peptides, proteins, nucleotides and nucleic acids, possess ionisable groups.
At any given pH, exist in solution as electrically charged species
1. Cations (+) : Positively charged ions
2. Anions (-) : Negatively charged ions
Under the influence of an electric field these charged particles will migrate either to the cathode or to the anode, depending on the nature of their net charge.
The protein molecules have a large number of ionizable amino and carboxyl groups on their surfaces.
The net charge density of the protein molecule which makes it move in an electric field in a direction and at a velocity dependent upon the sign and quantity of this net charge density.
It can be modified by solvents by increasing or decreasing ionic concentration of hydrogen ions.
Thus, if the acidity ofthe solvent is increased (H+ ions) the molecule will tend to become more positive and vice versa.
Even typically uncharged biomolecules such as carbohydrates can be made to wear weak charges through derivatization as borates and sometimes as phosphates.
Even if two molecules have the same charge, they might not migrate together because ifthere is difference in their molecular weights.
They will have different charge/mass ratio (this differences is of more use in electrophoresis on gels).
Taken together, these differences are sufficient to ensure differential migration when the ions in solution are subjected to an electric field.
This is the basis of electrophoresis.
MIGRATION OF AN ION IN AN ELECTRIC FIELD
A spherical molecule of net charge q is placed in an electric field.
The force Fwhich will act upon this particle will depend upon
(i) the net charge density of the molecule, and
(ii) the strength of the field in which it is placed.

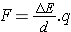 ........................................Equation 1
........................................Equation 1
whereIl ΔE/d is the field strength applied
(ΔE is the potential difference between the two electrodes, d being the distance between them).
The particle is in solution, hence the force acting can be given by Stoke's equation which takes in account of the extent of the friction.
The mobility depends upon
(i) the size and shape of the molecule
(ii) on the viscosity of the medium through which the molecule will migrate. :

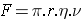 ........................................Equation 2 [Stoke's Equation]
........................................Equation 2 [Stoke's Equation]
Upon rearranging two equations

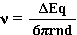
It will thus be seen that the velocity (v) of the molecule is
directly is proportional to
(i) the field strength (ΔE/d),
(ii) charge (q) on the molecule
inversely proportional to
(i) the particle size (6π).
(ii) viscosity of the solution (η)
toc | return to top | next page


