Water
Water is the most abundant substance in living systems, making up 70% or more of the weight of most organisms. The first living organisms arose in an aqueous environment. The course of evolution has been shaped by the properties of the aqueous medium in which life began.
Water comprises approximately 60–70 percent of the human body.
H2O is the molecular formula of water, also called Dihydrogen monoxide. Its IUPAC compliant name is Oxidane. H2O symbolises two hydrogen atoms and one oxygen atom. Its chemical formula H2O, indicates that each of its molecules contains one oxygen and two hydrogen atoms, connected by covalent bonds.
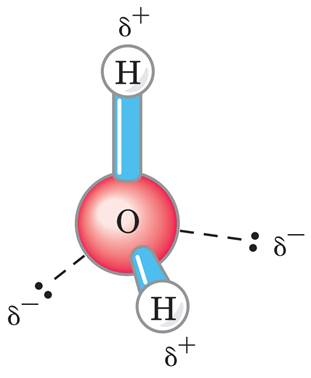
One of water's important properties is that it is composed of polar molecules. The two hydrogen atoms and one oxygen atom within water molecules (H2O) form polar covalent bonds. While there is no net charge to a water molecule, the polarity of water creates a slightly positive charge on hydrogen and a slightly negative charge on oxygen, contributing to water's properties of attraction. Water's charges are generated because oxygen is more electronegative, or electron loving, than hydrogen. Thus, it is more likely that a shared electron would be found near the oxygen nucleus than the hydrogen nucleus. Since water is a nonlinear, or bent, molecule, the difference in electronegativities between the oxygen and hydrogen atoms generates the partial negative charge near the oxygen and partial positive charges near both hydrogens.
https://bio.libretexts.org/Bookshelves/Introductory_and_General_Biology/Book%3A_General_Biology_(Boundless)/2%3A_The_Chemical_Foundation_of_Life/2.2%3A_Water/2.2A%3A_Waters_Polarity
Hydrogen bonds between water molecules provide the cohesive forces that make water a liquid at room temperature and that favor the extreme ordering of molecules that is typical of crystalline water (ice). Polar biomolecules dissolve readily in water because they can replace water-water interactions with more energetically favorable water-solute interactions. In contrast, nonpolar biomolecules interfere with water-water interactions but are unable to form water-solute interactions— consequently, nonpolar molecules are poorly soluble in water. In aqueous solutions, nonpolar molecules tend to cluster together.
Hydrogen bonds and ionic, hydrophobic (Greek, "water-fearing"), and van der Waals interactions are individually weak, but collectively they have a very significant influence on the three-dimensional structures of proteins, nucleic acids, polysaccharides, and membrane lipids.
Structure of the water molecule
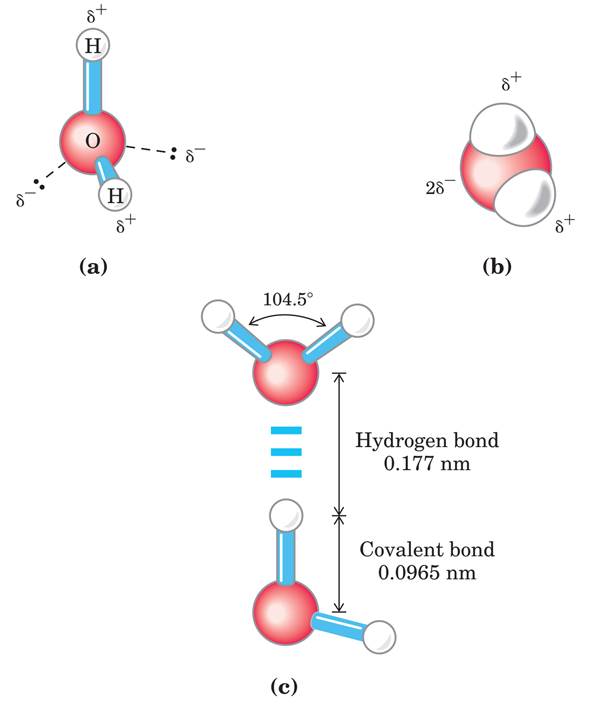
The dipolar nature of the H2O molecule is shown by (a) ball-and-stick and (b) space-filling models.
The dashed lines in (a) represent the nonbonding orbitals. There is a nearly tetrahedral arrangement of the outer-shell electron pairs around the oxygen atom; the two hydrogen atoms have localized partial positive charges (δ+) and the oxygen atom has a partial negative charge (2δ-).
(c) Two H2O molecules joined by a hydrogen bond (designated by three blue lines) between the oxygen atom of the upper molecule and a hydrogen atom of the lower one. Hydrogen bonds are longer and weaker than covalent O-H bonds.

