Hydrogen Bonding Gives Water Its Unusual Properties
Water has a
- higher Melting Point,
- Boiling point, and
- Heat of vaporization
than most other common solvents.
These unusual properties are a consequence of attractions between adjacent water molecules that give liquid water great internal cohesion.
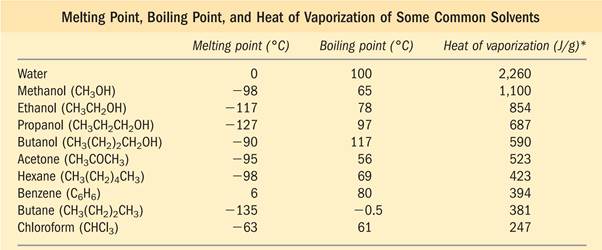
-
Polarity and Hydrogen Bonding:
- Water is a polar molecule due to its bent molecular structure and electronegativity difference between hydrogen and oxygen atoms.
- It forms strong hydrogen bonds between adjacent water molecules, resulting in high cohesion and surface tension.
-
Universal Solvent:
- Water's polarity allows it to dissolve a wide range of polar and ionic substances, making it a universal solvent in biological and chemical processes.
-
Heat Capacity:
- Water has a high specific heat capacity, meaning it can absorb and release large amounts of heat with minimal temperature change, stabilizing environments.
-
Heat of Vaporization:
- Water has a high heat of vaporization, requiring significant energy to evaporate, which is crucial for cooling organisms and regulating temperatures.
-
Density Anomaly:
- Water exhibits an unusual density anomaly where its solid form (ice) is less dense than its liquid form, due to the open arrangement of hydrogen bonds in ice.
-
Acid-Base Properties:
- Water can act as both an acid (donating a proton) and a base (accepting a proton) in reactions, playing a key role in acid-base chemistry and maintaining pH balance in biological systems.
-
Ionization:
- Water undergoes autoionization, forming hydronium (H3O+) and hydroxide (OH-) ions, contributing to its acidic and basic properties.
Biological Properties of Water:
-
Cellular Medium:
- Water is the primary constituent of cells and serves as a medium for biochemical reactions, allowing transport of nutrients, waste, and signaling molecules.
-
Hydration and Macromolecules:
- Water participates in the hydration of biomolecules like proteins, nucleic acids, and carbohydrates, influencing their structure and function.
-
Thermoregulation:
- Water's high heat capacity and heat of vaporization help organisms regulate body temperature through processes like sweating and evaporative cooling.
-
Transport Medium:
- Water acts as a transport medium in organisms, facilitating the movement of nutrients, gases (e.g., oxygen, carbon dioxide), and waste products.
-
Cohesion and Adhesion:
- Water's cohesive and adhesive properties enable capillary action, aiding in water transport through plants and maintaining blood flow in animals.
-
Ice Formation and Insulation:
- Ice formation on bodies of water acts as an insulating layer, protecting aquatic life beneath from extreme temperatures.
-
Chemical Reactions:
- Water participates in hydrolysis and dehydration reactions, essential for breaking down and building complex biomolecules.
-
Lubrication:
- Water acts as a lubricant in biological systems, reducing friction in joints and facilitating movement.
These properties of water play crucial roles in both chemical reactions and biological processes, contributing to the sustainability and complexity of life.

