Significance of Biological Buffers:
- Maintaining pH Homeostasis:
- Biological buffers play a crucial role in maintaining the pH of bodily fluids within a narrow range.
- This is essential because many biochemical processes are pH-dependent, and extreme pH levels can be harmful to cells and enzymes.
- Stabilizing Cellular Function:
- Buffers help stabilize intracellular and extracellular pH, ensuring that cellular processes function optimally.
- For example, enzymes and metabolic pathways have specific pH optima, and buffers help maintain these conditions.
- Preventing Acid-Base Imbalances:
- Biological buffers help prevent sudden shifts in pH that could lead to acidosis (low pH) or alkalosis (high pH), both of which can be life-threatening.
- They act as a first line of defense against changes in pH.
pH of Body Fluids like Blood and Saliva:
- Blood pH:
- Blood pH is tightly regulated within the range of 7.35 to 7.45.
- The primary buffer system in blood is the bicarbonate (HCO3-)/carbonic acid (H2CO3) system.
- It helps prevent significant changes in blood pH, even in the presence of metabolic acids or bases.
- Example: H2CO3 ⇌ HCO3- + H+, where H2CO3 is formed when CO2 dissolves in blood.
- Saliva pH:
- Saliva has a slightly acidic pH, typically around 6.5 to 7.5.
- It contains various buffers, including bicarbonate and phosphate, to maintain pH stability in the oral cavity.
- Saliva's pH helps with digestion, dental health, and protection against harmful microorganisms.
Blood Buffer Systems:
- Carbonate Buffer System:
- Bicarbonate (HCO3-) and carbonic acid (H2CO3) form a critical buffer system in blood.
- It regulates blood pH by converting excess H+ ions into H2O and CO2 or vice versa.
- Example: H2CO3 ⇌ HCO3- + H+.
- Acetate Buffer System:
- Acetate (CH3COO-) and acetic acid (CH3COOH) can function as blood buffers.
- They help maintain pH stability by reacting with excess H+ ions.
- Example: CH3COOH ⇌ CH3COO- + H+.
- Phosphate Buffer System:
- Phosphate ions (HPO4^2- and H2PO4-) are present in blood and can act as buffers.
- They are especially important in maintaining pH within cells and urine.
- Example: H2PO4- ⇌ HPO42- + H+.
Protein Buffers:
- Buffering Proteins:
- Proteins, such as hemoglobin and albumin, can act as buffers in blood and other bodily fluids.
- Their amino acid residues can accept or release H+ ions, regulating pH.
- For example, histidine residues in hemoglobin can buffer blood pH.
- Hemoglobin as a Buffer:
- Hemoglobin, the oxygen-carrying protein in red blood cells, can also buffer blood pH.
- When oxygen binds to hemoglobin, it increases its affinity for H+ ions, acting as a buffer in the presence of acidic conditions.
Significance of TRIS Buffers :
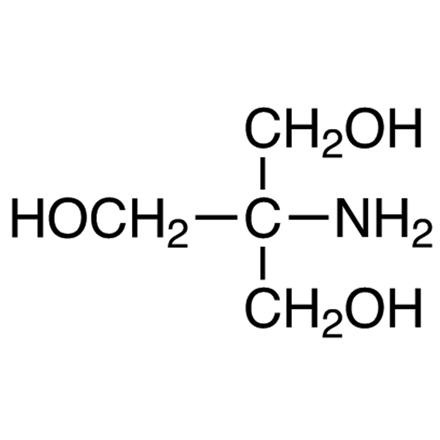
- TRIS Buffer:
- TRIS (Tris(hydroxymethyl)aminomethane) is a commonly used buffer in biochemical and molecular biology experiments.
- It has a pKa of around 8.1, making it effective in the physiological pH range.
- TRIS buffers are known for their stability and minimal interference with biochemical reactions.
- Applications of TRIS Buffers:
- TRIS buffers are used in various laboratory techniques, including DNA and protein electrophoresis, enzyme assays, and protein purification.
- They help maintain a constant pH, ensuring accurate and reproducible results in experiments.
- Example: A 0.1 M TRIS-HCl buffer at pH 7.4 is often used in cell culture media for biological research.

