Carbohydrates
It is the most abundant biomolecules on Earth. Each year, photosynthesis converts more than 100 billion metric tons of CO2 and H2O into cellulose and other plant products.
The term 'carbohydrate' was originally coined for this class of compounds as most of them were 'hydrates of carbon' or could be represented by the general formula,
Cx(H2O)y.
Later, it was found that some of them, such as deoxyribose (C5H10O4) and rhamnose (C6H12O5) do not have the required ratio of hydrogen to oxygen. In addition, certain other carbohydrates are now known to possess nitrogen (e.g., glucosamine, C6H13O5N), phosphorus or sulfur also and obviously do not coincide with the above general formula.
Furthermore, formaldehyde (H.CHO or CH2O), acetic acid (CH3.COOH or C2H4O2) and lactic acid (CH3.CHOH.COOH or C3H6O3) which have C, H and O and the ratio of H : O is also the same as in water, but are not a carbohydrates. Hence, the continued usage of the term 'carbohydrate' is for convenience rather than exactness.
"The carbohydrates are defined as polyhydroxy aldehydes or ketones and their derivatives or as substances that yield one of these compounds on hydrolysis."
Functions of carbohydrates
Carbohydrates participate in a wide range of functions
1. They are the most abundant dietary source of energy (4 Cal/g) for all organisms.
2. Carbohydrates are precursors for many organic compounds (fats, amino acids).
3. Carbohydrates (as glycoproteins and glycolipids) participate in the structure of cell membrane and cellular functions such as cell growth, adhesion and fertilization.
4. They are structural components of many organisms. These include the fiber (cellulose) of plants, exoskeleton of some insects and the cell wall of microorganisms.
5. Carbohydrates also serve as the storage form of energy (glycogen) to meet the immediate energy demands of the body.
CLASSIFICATION OF CARBOHYDRATES
Carbohydrates can be classified into three groups based on the number of sugar units they contain:
1. Monosaccharides,
2. Oligosaccharides, and
3. Polysaccharides.
Monosaccharides
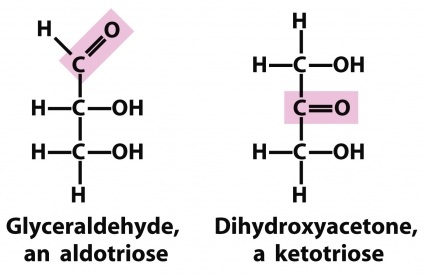
They are simple sugars that cannot be hydrolyzed into smaller units under reasonably mild conditions.
The simplest monosaccharides fitting our definition and empirical formula are the aldose glyceraldehyde and its isomer, the ketose dihydroxy acetone. Both of these sugars are trioses because they contain three carbon atoms. In addition to the functional groups (aldehyde and ketone) that are used in describing these two sugars, note that they contain alcoholic hydroxyls and, in the case of glyceraldehyde, an asymmetric carbon atom.
Oligosaccharides
They are hydrolyzable polymers of monosaccharides that contain from two to six molecules of simple sugars.
The disaccharides, which have two monosaccharide units, are the most abundant; trisaccharides also occur free in nature.
Oligosaccharides with more than three subunits are usually found bound as side chains in glycoproteins.
Polysaccharides
They are polymers, frequently insoluble, consisting of hundreds or thousandsof monosaccharide units; they may beeither linear or branched in structure.
If the polymer is made up from a single monosaccharide, the polysaccharide is called a homopolysaccharide.
If two or more different monosaccharides are found in the polymer, it is called a heteropolysaccharide.
Some of the monosaccharides that are bound together by glycosidic bondsto form polysaccharides are glucose, xylose, and arabinose.

