Optical activity of sugars
Optical activity is a characteristic feature of compounds with asymmetric carbon atom.
When a beam of polarized light is passed through a solution of an optical isomer, it will be rotated either to the right or left.
The term dextrorotatory (d+) and levorotatory (l–) are used to compounds that respectively rotate the plane of polarized light to the right or to the left.
An optical isomer may be designated as D(+), D(–), L(+) and L(–) based on its structural relation with glyceraldehyde. It may be noted that the D- and L-configurations of sugars are primarily based on the structure of glyceraldehyde, the optical activities however, may be different.
Racemic mixture :
If d- and l-isomers are present in equal concentration, it is known as racemic mixture or dl mixture.
Racemic mixture does not exhibit any optical activity, since the dextro- and levorotatory activities cancel each other.
In the medical practice, the term dextrose is used for glucose in solution. This is because of the dextrorotatory nature of glucose.
Configuration of D-aldoses
The configuration of possible D-aldoses starting from D-glyceraldehyde is depicted in Fig..
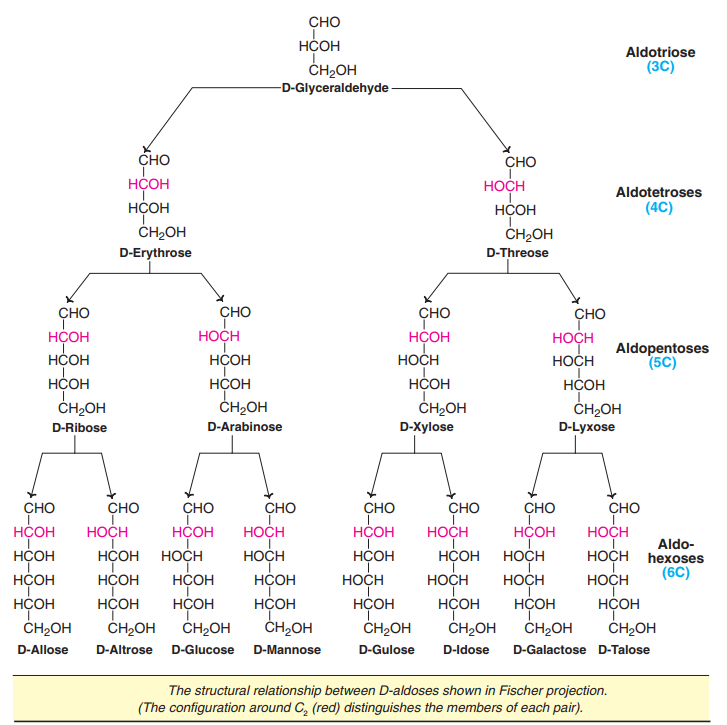
This is a representation of KillianiFischer synthesis, by increasing the chain length of an aldose, by one carbon at a time. Thus, starting with an aldotriose (3C), aldotetroses (4C), aldopentoses (5C) and aldohexoses (6C) are formed. Of the 8 aldohexoses, glucose, mannose and galactose are the most familiar. Among these, D-glucose is the only aldose monosaccharide that predominantly occurs in nature.
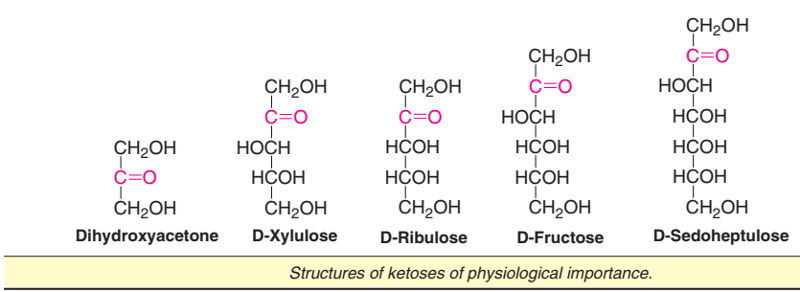
Configuration of D-ketoses
Starting from dihydroxyacetone (triose), there are five keto-sugars which are physiologically important. Their structures are given in Fig.
Epimers
If two monosaccharides differ from each other in their configuration around a single specific carbon (other than anomeric) atom, they are referred to as epimers to each other (Fig.).
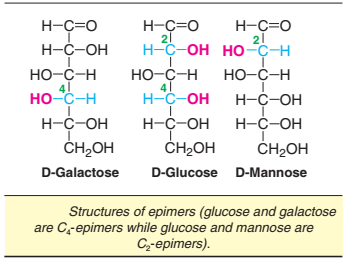
For instance, glucose and galactose are epimers with regard to carbon 4 (C4-epimers). That is, they differ in the arrangement of OH group at C4. Glucose and mannose are epimers with regard to carbon 2 (C2-epimers). The interconversion of epimers (e.g. glucose to galactose and vice versa) is known as epimerization, and a group of enzymes—namely—epimerases catalyse this reaction.
Enantiomers
Enantiomers are a special type of stereoisomers that are mirror images of each other. The two members are designated as D- and L-sugars. Enantiomers of glucose are depicted in Fig.
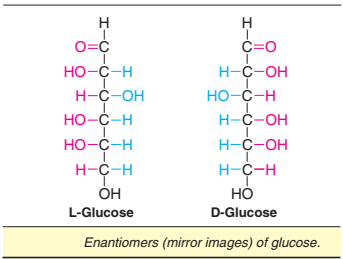
Majority of the sugars in the higher animals (including man) are of D-type. The term diastereomers is used to represent the stereoisomers that are not mirror images of one another.
