REACTIONS OF MONOSACCHARIDES
Tautomerization or enolization
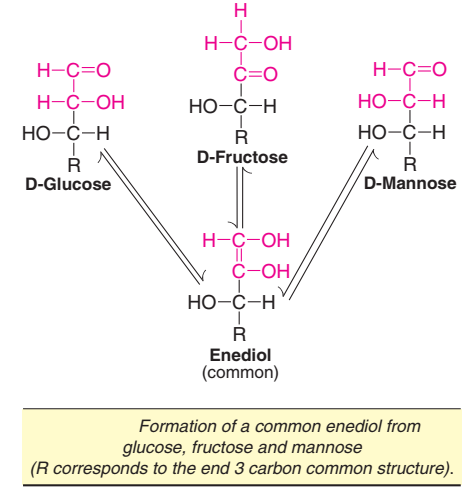
The process of shifting a hydrogen atom from one carbon atom to another to produce enediols is known as tautomerization. Sugars possessing anomeric carbon atom undergo tautomerization in alkaline solutions. When glucose is kept in alkaline solution for several hours, it undergoes isomerization to form D-fructose and D-mannose. This reaction— known as the Lobry de Bruyn-von Ekenstein transformation—results in the formation of a common intermediate—namely enediol—for all the three sugars, as depicted in Fig. The enediols are highly reactive, hence sugars in alkaline solution are powerful reducing agents.
Reducing properties
The sugars are classified as reducing or nonreducing. The reducing property is attributed to the free aldehyde or keto group of anomeric carbon.
In the laboratory, many tests are employed to identify the reducing action of sugars. These include Benedict's test, Fehling's test, Barfoed's test etc. The reduction is much more efficient in the alkaline medium than in the acid medium.
The enediol forms (explained above) or sugars reduce cupric ions (Cu2+) of copper sulphate to cuprous ions (Cu+), which form a yellow precipitate of cuprous hydroxide or a red precipitate of cuprous oxide as shown next. It may be noted that the reducing property of sugars cannot help for a specific identification of any one sugar, since it is a general reaction.
Oxidation
Depending on the oxidizing agent used, the terminal aldehyde (or keto) or the terminal alcohol or both the groups may be oxidized. For instance, consider glucose :
1. Oxidation of aldehyde group (CHO → COOH) results in the formation of gluconic acid.
2. Oxidation of terminal alcohol group (CH2OH → COOH) leads to the production of glucuronic acid.
Reduction
When treated with reducing agents such as sodium amalgam, the aldehyde or keto group of monosaccharide is reduced to corresponding alcohol, as indicated by the general formula :
