STRUCTURE OF GLUCOSE
For a better understanding of glucose structure, let us consider the formation of hemiacetals and hemiketals, respectively produced when an aldehyde or a ketone reacts with alcohol.
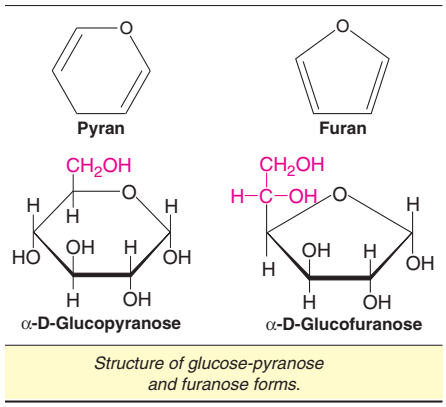
The aldehyde group of glucose at C1 reacts with alcohol group at C5 to form two types of cyclic hemiacetals namely D and E, as depicted.
Pyranose and furanose structures
Haworth projection formulae are depicted by a six-membered ring pyranose (based on pyran) or a five-membered ring furanose (based on furan). The cyclic forms of glucose are known as α-D-glucopyranose and α-D-glucofuranose.
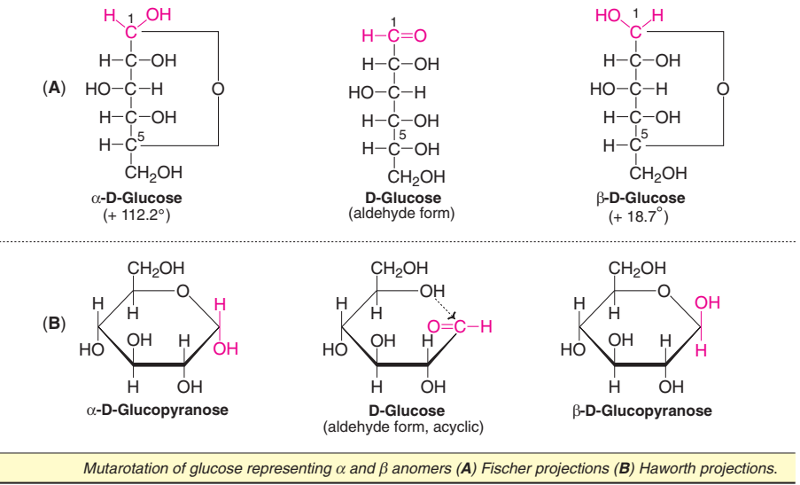
Anomers—mutarotation
The α and β cyclic forms of D-glucose are known as anomers. They differ from each other in the configuration only around C1 known as anomeric carbon (hemiacetal carbon).
In case of α anomer, the -OH group held by anomeric carbon is on the opposite side of the group -CH2OH of sugar ring. The reverse is true for β-anomer. The anomers differ in certain physical and chemical properties.
Mutarotation :
The α and β anomers of glucose have different optical rotations. The specific optical rotation of a freshly prepared glucose (α anomer) solution in water is +112.2° which gradually changes and attains an equilibrium with a constant value of +52.7°. In the presence of alkali, the decrease in optical rotation is rapid. The optical rotation of β-glucose is +18.7°. Mutarotation is defined as the change in the specific optical rotation representing the interconversion of α and β forms of D-glucose to an equilibrium mixture.

The equilibrium mixture contains 63% β-anomer and 36% α-anomer of glucose with 1% open chain form. In aqueous solution, the β form is more predominant due to its stable conformation. The α and β forms of glucose are interconvertible which occurs through a linear form. The latter, as such, is present in an insignificant quantity.
Mutarotation of fructose : Fructose also exhibits mutarotation. In case of fructose, the pyranose ring (six-membered) is converted to furanose (five-membered) ring, till an equilibrium is attained. And fructose has a specific optical rotation of –92° at equilibrium.
