TRANSAMINATION
The transfer of an amino ( NH2) group from an amino acid to a keto acid is known as transamination.
This process involves the interconversion of a pair of amino acids and a pair of keto acids, catalysed by a group of enzymes called transaminases (recently, aminotransferases).
Salient features of transamination
1. All transaminases require pyridoxal phosphate (PLP), a coenzyme derived from vitamin B6.
2. Specific transaminases exist for each pair of amino and keto acids. However, only two— namely, aspartate transaminase and alanine transaminase—make a significant contribution for transamination.
3. There is no free NH3 liberated, only the transfer of amino group occurs.
4. Transamination is reversible [see below]
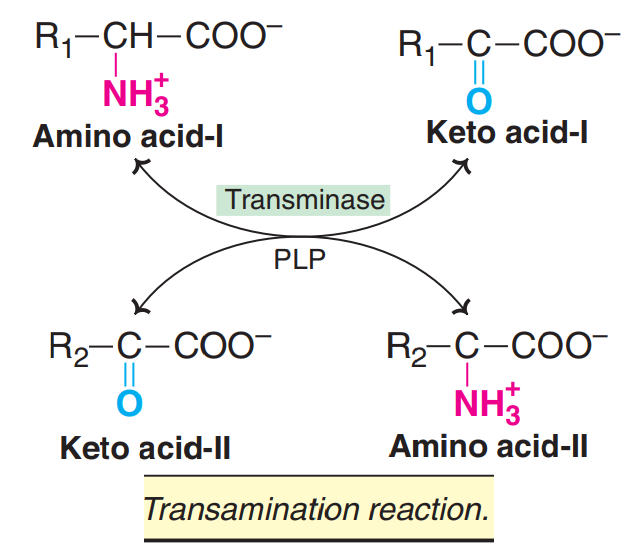
5. Transamination is very important for the redistribution of amino groups and production of non-essential amino acids, as per the requirement of the cell. It involves both catabolism (degradation) and anabolism (synthesis) of amino acids.
6. Transamination diverts the excess amino acids towards energy generation.
7. The amino acids undergo transamination to finally concentrate nitrogen in glutamate. Glutamate is the only amino acid that undergoes oxidative deamination to a significant extent to liberate free NH3 for urea synthesis.
8. All amino acids except lysine, threonine, proline and hydroxyproline participate in transamination.
9. Transamination is not restricted to D-amino groups only. For instance, G-amino group of ornithine is transaminated.
10. Serum transaminases are important for diagnostic and prognostic purposes.
Mechanism of transamination
Transamination occurs in two stages
1. Transfer of the amino group to the coenzyme pyridoxal phosphate (bound to the coenzyme) to form pyridoxamine phosphate.
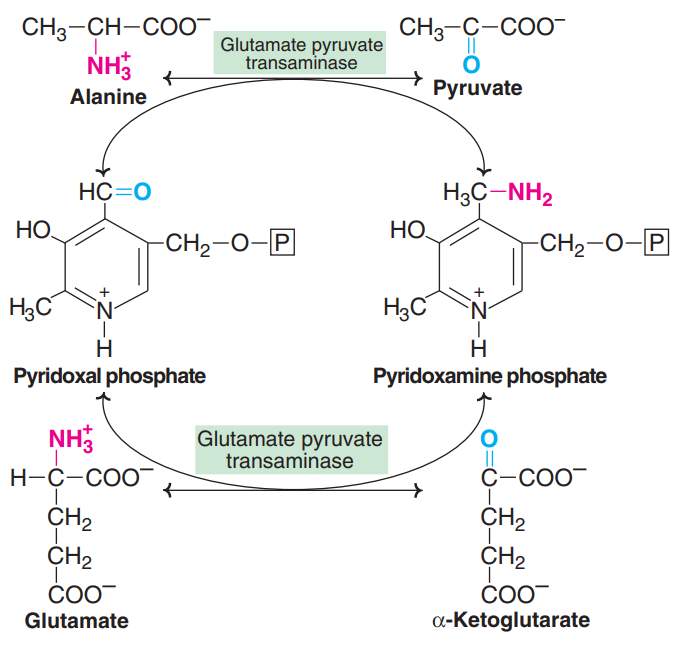
2. The amino group of pyridoxamine phosphate is then transferred to a keto acid to produce a new amino acid and the enzyme with PLP is regenerated.
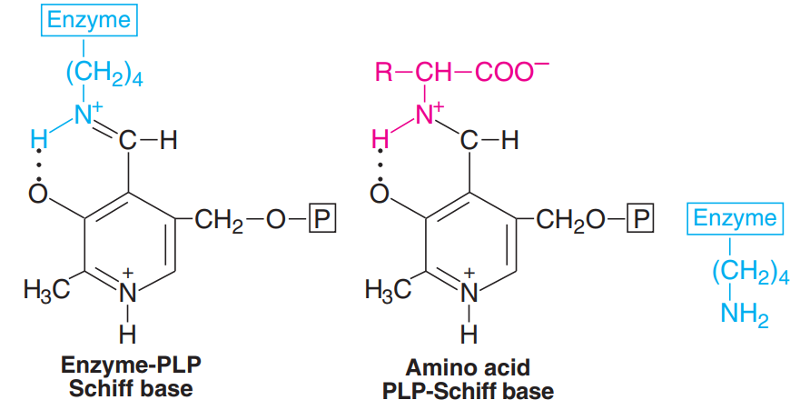
All the transaminases require pyridoxal phosphate (PLP), a derivative of vitamin B6.
The aldehyde group of PLP is linked with H-amino group of lysine residue, at the active site of the enzyme forming a Schiff base (imine linkage).
When an amino acid (substrate) comes in contact with the enzyme, it displaces lysine and a new Schiff base linkage is formed.
The amino acid-PLP-Schiff base tightly binds with the enzyme by noncovalent forces.
Snell and Braustein proposed a Ping Pong Bi Bi mechanism involving a series of intermediates (aldimines and ketimines) in transamination reaction.
