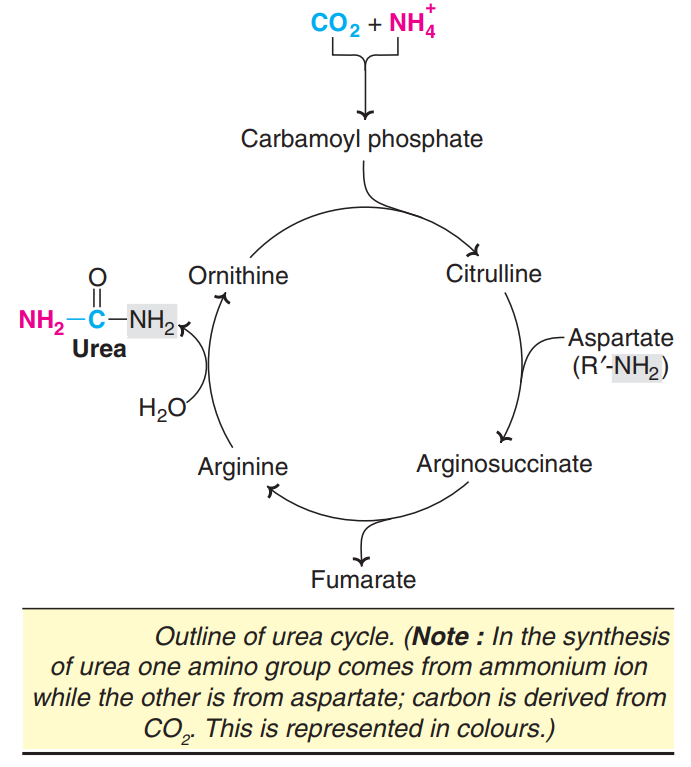
UREA CYCLE
Urea is the end product of protein metabolism (amino acid metabolism).
The nitrogen of amino acids, converted to ammonia, is toxic to the body. It is converted to urea and detoxified. As such, urea accounts for 80-90% of the nitrogen containing substances excreted in urine.
Urea is synthesized in liver and transported to kidneys for excretion in urine.
Urea cycle is the first metabolic cycle that was elucidated by Hans Krebs and Kurt Henseleit (1932), hence it is known as Krebs-Henseleit cycle.
The individual reactions, however, were described in more detail later on by Ratner and Cohen.
Urea has two amino ( NH2) groups, one derived from NH3 and the other from aspartate. Carbon atom is supplied by CO2.
Urea synthesis is a five-step cyclic process, with five distinct enzymes.
The first two enzymes are present in mitochondria while the rest are localized in cytosol.
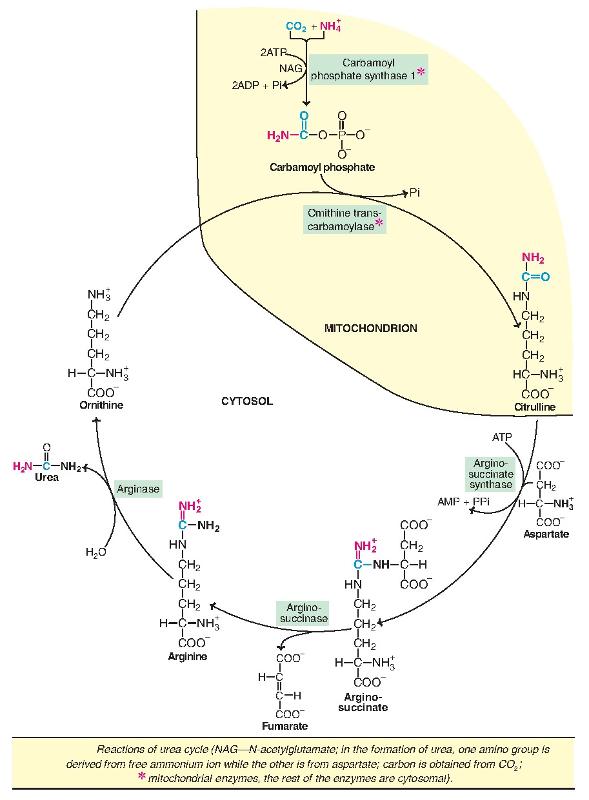
1. Synthesis of carbamoyl phosphate :
Carbamoyl phosphate synthase I (CPS I) of mitochondria catalyses the condensation of NH4+ ions with CO2 to form carbamoyl phosphate.
This step consumes two ATP and is irreversible, and rate-limiting. CPS I requires Nacetylglutamate for its activity. Another enzyme, carbamoyl phosphate synthase II (CPS II)— involved in pyrimidine synthesis—is present in cytosol.
It accepts amino group from glutamine and does not require N-acetylglutamate for its activity.
2. Formation of citrulline :
Citrulline is synthesized from carbamoyl phosphate and ornithine by ornithine transcarbamoylase. Ornithine is regenerated and used in urea cycle. Therefore, its role is comparable to that of oxaloacetate in citric acid cycle. Ornithine and citrulline are basic amino acids. (They are never found in protein structure due to lack of codons). Citrulline produced in this reaction is transported to cytosol by a transporter system.
3. Synthesis of arginosuccinate :
Arginosuccinate synthase condenses citrulline with aspartate to produce arginosuccinate. The second amino group of urea is incorporated in this reaction. This step requires ATP which is cleaved to AMP and pyrophosphate (PPi). The latter is immediately broken down to inorganic phosphate (Pi).
4. Cleavage of arginosuccinate :
Arginosuccinase cleaves arginosuccinate to give arginine and fumarate. Arginine is the immediate precursor for urea. Fumarate liberated here provides a connecting link with TCA cycle, gluconeogenesis etc.
5. Formation of urea :
Arginase is the fifth and final enzyme that cleaves arginine to yield urea and ornithine. Ornithine, so regenerated, enters mitochondria for its reuse in the urea cycle. Arginase is activated by Co2+ and Mn2+. Ornithine and lysine compete with arginine (competitive inhibition). Arginase is mostly found in the liver, while the rest of the enzymes (four) of urea cycle are also present in other tissues. For this reason, arginine synthesis may occur to varying degrees in many tissues. But only the liver can ultimately produce urea.
Overall reaction and energetics
The urea cycle is irreversible and consumes 4 ATP. Two ATP are utilized for the synthesis of carbamoyl phosphate. One ATP is converted to AMP and PPi to produce arginosuccinate which equals to 2 ATP. Hence 4 ATP are actually consumed.
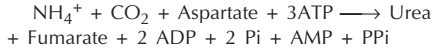
Regulation of urea cycle
The first reaction catalysed by carbamoyl phosphate synthase I (CPS I) is rate-limiting reaction or committed step in urea synthesis.
CPS I is allosterically activated by N-acetylglutamate (NAG).
It is synthesized from glutamate and acetyl CoA by synthase and degraded by a hydrolase.
The rate of urea synthesis in liver is correlated with the concentration of N-acetylglutamate.
High concentrations of arginine increase NAG. The consumption of a protein-rich meal increases the level of NAG in liver, leading to enhanced urea synthesis.
Carbamoyl phosphate synthase I and glutamate dehydrogenase are localized in the mitochondria. They coordinate with each other in the formation of NH3, and its utilization for the synthesis of carbamoyl phosphate.
The remaining four enzymes of urea cycle are mostly controlled by the concentration of their respective substrates.
Disposal of urea
Urea produced in the liver freely diffuses and is transported in blood to kidneys, and excreted.
A small amount of urea enters the intestine where it is broken down to CO2 and NH3 by the bacterial enzyme urease.
This ammonia is either lost in the feces or absorbed into the blood.
In renal failure, the blood urea level is elevated (uremia), resulting in diffusion of more urea into intestine and its breakdown to NH3.
Hyperammonemia (increased blood NH3) is commonly seen in patients of kidney failure. For these patients, oral administration of antibiotics (neomycin) to kill intestinal bacteria is advised.
Integration between urea cycle and TCA cycle
Urea cycle is linked with TCA cycle in three different ways. This is regarded as bicyclic integration between the two cycles.
1. The production of fumarate in urea cycle
It is the most important integrating point with TCA cycle. Fumarate is converted to malate and then to oxaloacetate in TCA cycle. Oxaloacetate undergoes transamination to produce aspartate which enters urea cycle. Here, it combines with citrulline to produce arginosuccinate. Oxaloacetate is an important metabolite which can combine with acetyl CoA to form citrate and get finally oxidized. Oxaloacetate can also serve as a precursor for the synthesis of glucose (gluconeogenesis).
2. ATP
ATP (12) are generated in the TCA cycle while ATP (4) are utilized for urea synthesis.
3. CO2
Citric acid cycle is an important metabolic pathway for the complete oxidation of various metabolites to CO2 and H2O. The CO2 liberated in TCA cycle (in the mitochondria) can be utilized in urea cycle.
