DEAMINATION
The removal of amino group from the amino acids as NH3 is deamination.
The deamination results in the liberation of ammonia for urea synthesis. Simultaneously, the carbon skeleton of amino acids is converted to keto acids.
Deamination may be either oxidative or non-oxidative.
Although transamination and deamination are separately discussed, they occur simultaneously, often involving glutamate as the central molecule. For this reason, some authors use the term transdeamination while describing the reactions of transamination and deamination, particularly involving glutamate.
I. Oxidative deamination
Oxidative deamination is the liberation of free ammonia from the amino group of amino acids coupled with oxidation.
This takes place mostly in liver and kidney.
The purpose of oxidative deamination is to provide NH3 for urea synthesis and D-keto acids for a variety of reactions, including energy generation.
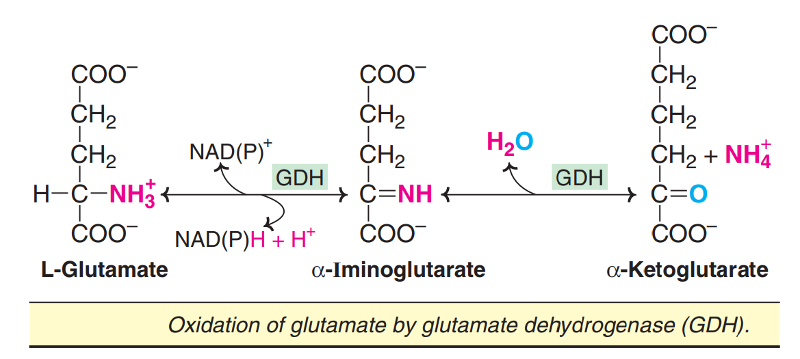
In the process of transamination, the amino groups of most amino acids are transferred to D-ketoglutarate to produce glutamate. Thus, glutamate serves as a 'collection centre' for amino groups in the biological system. Glutamate rapidly undergoes oxidative deamination, catalysed by glutamate dehydrogenase (GDH) to liberate ammonia.
This enzyme is unique in that it can utilize either NAD+ or NADP+ as a coenzyme.
Conversion of glutamate to D-ketoglutarate occurs through the formation of an intermediate, D-iminoglutarate.
Glutamate dehydrogenase catalysed reaction is important as it reversibly links up glutamate metabolism with TCA cycle through D-ketoglutarate.
GDH is involved in both catabolic and anabolic reactions.
Regulation of GDH activity :
Glutamate dehydrogenase is a zinc containing mitochondrial enzyme.
It is a complex enzyme consisting of six identical units with a molecular weight of 56,000 each. GDH is controlled by allosteric regulation. GTP and ATP inhibit—whereas GDP and ADP activate—glutamate dehydrogenase. Steroid and thyroid hormones inhibit GDH.
After ingestion of a protein-rich meal, liver glutamate level is elevated. It is converted to D-ketoglutarate with liberation of NH3.
Further, when the cellular energy levels are low, the degradation of glutamate is increased to provide D-ketoglutarate which enters TCA cycle to liberate energy.
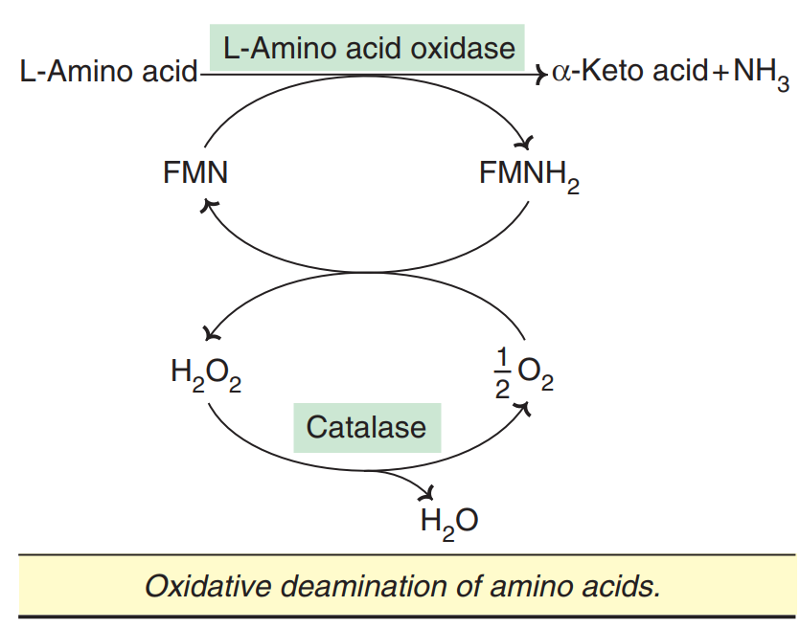
Oxidative deamination by amino acid oxidases :
L-Amino acid oxidase and D-amino acid oxidase are flavoproteins, possessing FMN and FAD, respectively.
They act on the corresponding amino acids (L or D) to produce D-keto acids and NH3.
In this reaction, oxygen is reduced to H2O2, which is later decomposed by catalase.
Fate of D-amino acids :
D-Amino acids are found in plants and microorganisms. They are, however, not present in the mammalian proteins.
But D-amino acids are regularly taken in the diet and metabolized by the body.
D-Amino acid oxidase converts them to the respective D-keto acids by oxidative deamination.
The D-keto acids so produced undergo transamination to be converted to L-amino acids which participate in various metabolisms.
Keto acids may be oxidized to generate energy or serve as precursors for glucose and fat synthesis.
Thus, D-amino acid oxidase is important as it initiates the first step for the conversion of unnatural D-amino acids to L-amino acids in the body.
II. Non-oxidative deamination
Some of the amino acids can be deaminated to liberate NH3 without undergoing oxidation
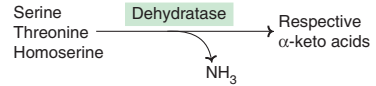
(a) Amino acid dehydrases :
Serine, threonine and homoserine are the hydroxy amino acids.
They undergo non-oxidative deamination catalysed by PLP-dependent dehydrases (dehydratases).
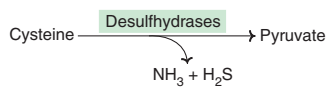
(b) Amino acid desulfhydrases :
The sulfur amino acids, namely cysteine and homocysteine, undergo deamination coupled with desulfhydration to give keto acids.
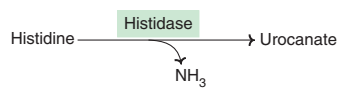
(c) Deamination of histidine :
The enzyme histidase acts on histidine to liberate NH3 by a non-oxidative deamination process.