III. Terminal Oxidation (Electron Transport System or Respiratory Chain):
The oxidation of reduced coenzymes into their oxidized forms is known as terminal oxidation.
This process involves electron transfer through various electron carriers which form electron transport (transfer) system or respiratory chain.
Free molecular oxygen is the final acceptor of electrons.
During this process, synthesis of ATP takes place with the help of energy released during the electron transfer.
This synthesis of ATP is called oxidative phosphorylation.
During glycolysis and Krebs cycle, respiratory substrates are oxidized at several steps.
It occurs by removal of hydrogen (dehydrogenation).
This hydrogen is accepted by coenzymes - mostly NAD and sometimes FAD so that they are reduced to NADH2 and FADH2 respectively.
Such reduced coenzymes are converted back to their oxidized forms.
The hydrogen removed from reduced coenzymes is released into the matrix of mitochondria during the process.
This H2 reacts with atmospheric oxygen to form water molecule. This is called metabolic water and is formed at the end of electron transport system (ETS), and hence is called terminal oxidation.
The electron transport system (ETS) includes
- FMN as the co-enzyme, Co-enzyme-Q (or ubiquinone) as both a coenzyme
- electron carrier and a number of electron carriers called cytochromes. They include cytochromes b, c c, a and a3.
- Cytochromes are iron containing compounds. Iron keeps oscillating between ferric and ferrous forms.
When electrons (2e") are taken up by the cytochrome, ferric compound gets reduced to ferrous and as it gives the electrons to the next cytochrome in the chain it gets reoxidized to ferric.
- The final electron acceptor is atmospheric oxygen.
Various electron carriers are arranged on the body of the oxysomes in the order of their decreasing energy level, NAD being the first and oxygen the last.
Thus, NAD has the highest energy while oxygen has the lowest energy. When a reduced coenzyme such as NADH2 or FADH2 from glycolysis or Krebs cycle, enters the respiratory chain, hydrogen is removed and NAD or FAD as the case may be, is regenerated.
The hydrogen further splits into proton and an electron. (H+ and e-).
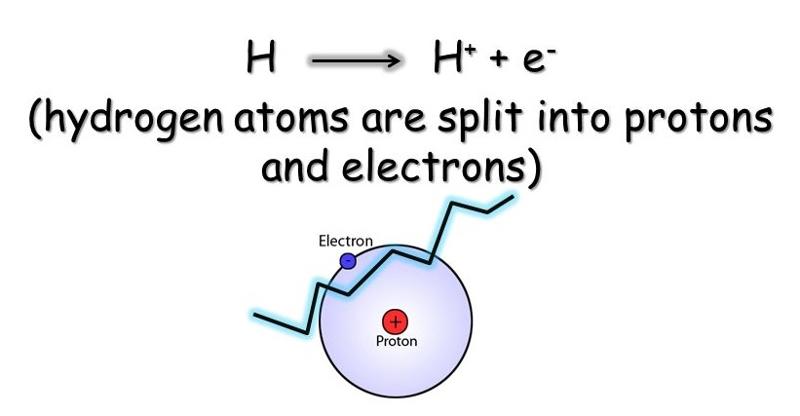
The electrons pass through the electron transport system so that the electron acceptors get oxidized and reduced alternately.
They move along FMN, Co-Q, cytochrome b, p., c, a and a .
The protons are released in the mitochondrial matrix.
Finally, the two electrons react with atmospheric oxygen to produce O ' which joins with 2H+ and produce a water molecule.
This is called metabolic water. When the electrons are being transported along the electron carriers, energy is released during each transfer.
At certain stages, it is utilized for synthesis of ATP from ADP and Pi.
The biosynthesis of ATP molecule using energy released during oxidation of respiratory substrates is called oxidative phosphorylation.
Three ATP molecules are synthesized through ETS during electron transfer as shown in Fig.
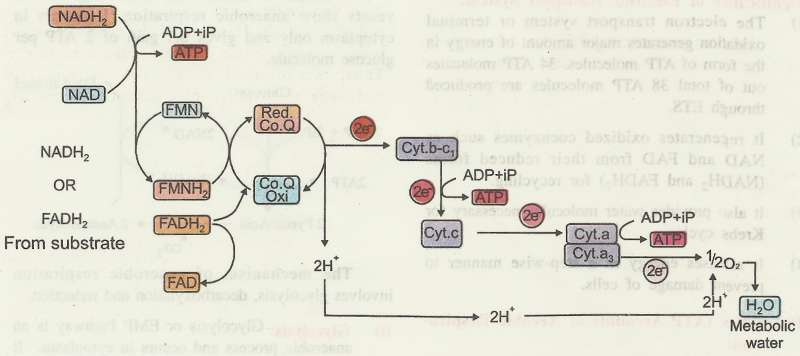
- first ATP molecule is formed between NADH2 and FMN,
- second ATP between cytochrome complex - bc1 and cytochrome c, and
- the third ATP between cytochrome a and cytochrome a3
Thus,
- when NADH2 is reoxidized, three ATP molecules are formed and
- when FADH2 is reoxidized, only two ATP molecules are formed.
Significance of Electron Transport System:
1) The electron transport system or terminal oxidation generates major amount of energy in the form of ATP molecules.
34 ATP molecules out of total 38 ATP molecules are produced through ETS.
2) It regenerates oxidized coenzymes such as NAD and FAD from their reduced forms (NADH2 and FADH2) for recycling.
3) It also provides water molecules necessary for Krebs cycle.
4) It releases energy in a step-wise manner to prevent damage of cells.
