Classification of Enzymes :
1. Oxidoreductases :
These are enzymes catalyzing oxidation and reduction reactions by the transfer of hydrogen and/or oxygen, e.g. alcohol dehydrogenase
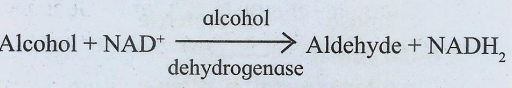
2. Transferases :
These enzymes catalyse he transfer of certain groups between two molecules, e.g. glucokinase.
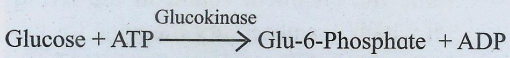
3. Hydrolases:
These are enzymes catalyse hydrolytic reactions. This class includes amylases, proteases, lipases etc. eg. Sucrase
![]()
4. Lyases :
These enzymes are involved in elimination reactions resulting in the removal of a group of atoms from substrate molecule to leave a double bond. It includes aldolases, decarboxylases, and dehydratases, e.g fumarate hydratase.
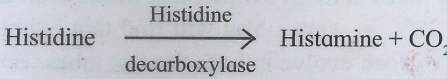
5. Isomerases
These enzymes catalyze structural rearrangements within a molecule, Their nomenclature is based on the type of isomerism.Thus these enzymes are identified as racemases, epimerases, isomerases, mutases, e.g. xylose isomerase.
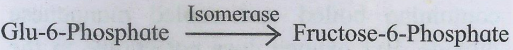
6. Ligases or Synthetases :
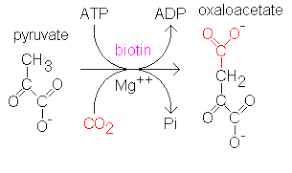
These are the enzymes which catalyse the covalent linkage of the molecules utilizing the energy obtained from hydrolysis of an energy-rich compound like ATP, GTP e.g. glutathione synthetase, Pyruvate carboxylase.
??Can you tell?
1. Which enzyme is needed to digest food reserve in castor seed?
a. Amylase b. Diastase c. Lipase d. protease
2. Co-enzyme is
a often a metal
b. often a vitamin
c. always as organic molecule
d. always an inorganic molecule
3. List the important properties of enzymes.
4. Name the chemical found in the living cell which has necessary message for the production of all enzymes required by it.
Try This:
To demonstrate the effect of heat on the activities of inorganic catalysts and enzymes.
Take 2 ml of hydrogen peroxide (H202) in two test tubes, Add a pinch of manganese dioxide (Mn02) powder to one and a small piece of potato (to provide enzyme catalase) or fresh liver (to provide enzyme peroxidase) to other test tube. Keep the tubes at room temperature in summer and at 38°C in winter. You will find that bubbles of oxygen evolve in both the test tubes. Both Mn02 and cellular enzymes (catalase or peroxidase) cause breakdown of H2O2 and evolution of oxygen. Now take two fresh test tubes and repeat the experiment. This time, use boiled and cooled manganese dioxide and the liver/potato piece. You will find that oxygen evolves in the hydrogen peroxide solution containing boiled and cooled manganese dioxide. But oxygen does not evolve in the other tube containing boiled and cooled liver/ potato piece. This activity confirms that heat does not affect catalytic action of inorganic catalyst but inactivates the enzyme.
