CHEMICAL BOND - ATTAINMENT OF A STABLE CONFIGURATION
The most stable electronic arrangement is a noble gas structure.
Many molecules have this arrangement.
Less stable arrangements than this are commonly attained by transition elements.
Valence
The term valence (or valency) is often used to state the potential or capacity of an element to combine with other elements.
Valence is the number of bonds formed by an atom in a molecule.
In hydrogen chloride (HCl), one atom of chlorine is combined with one atom of hydrogen and the valence of chlorine is 1.
In magnesium oxide (MgO), since one atom of magnesium holds one atom of oxygen, the valence of magnesium is 2.
By the above definition, we would assign a valence of 2 to sulphur in H2S, but 4 to sulphur in SO2.
Some elements have fractional valence in certain compounds, while there are elements that have variable valencies.
The concept of valence as a mere number could not explain these facts.
This concept, in fact, was very confusing and has lost all value.
Valence Electrons
The electrons in the outer energy level of an atom are the ones that can take part in chemical bonding.
These electrons are, therefore, referred to as the valence electrons.
The electronic configuration of Na is 2, 8, 1 and that of Cl is 2, 8, 7.
Thus sodium has one valence electron and chlorine 7.
It is important to remember that for an A group element of the periodic table (H, O, K, F, Al etc.) the group number is equal to the number of valence electrons.
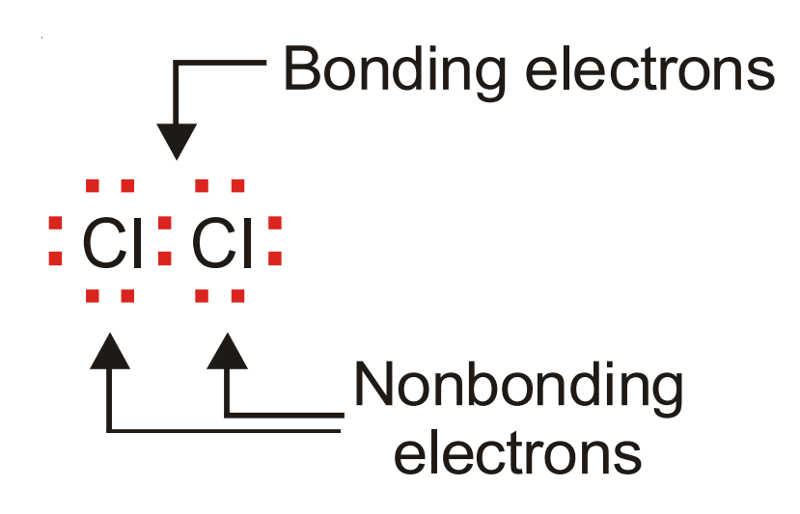
Bonding and Non-bonding Electrons
The valence electrons actually involved in bond formation are called bonding electrons.
The remaining valence electrons still available for bond formation are referred to as non-bonding electrons.
Thus :
Lewis Symbols of Elements
A Lewis symbol of an element consists of an element's symbol and surrounding dots to represent the number of valence electrons.
In this notation, the symbol of an element represents the nucleus plus the inner normally filled levels (or shells) of the atom.
For illustration, the symbol Na stands for the nucleus of sodium atom plus 2, 8 electrons in the inner two levels.
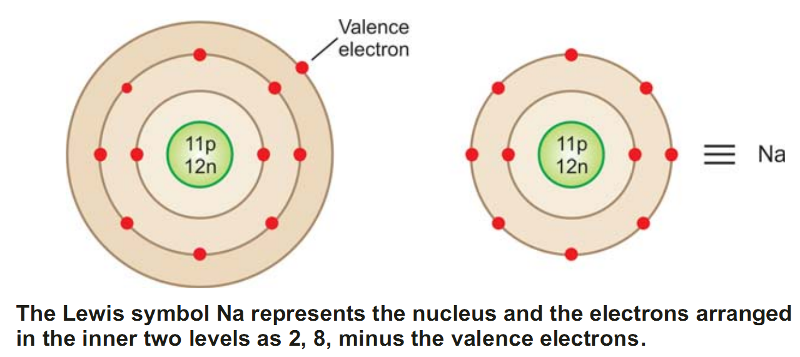
To represent a Lewis symbol for an element, write down the symbol of the element and surround the symbol with a number of dots (or crosses) equal to the number of valence electrons.
The position of dots around the symbol is not really of any significance.
The bonding electrons are shown at appropriate positions, while the rest of the electrons are generally given in pairs.
The Lewis symbols for hydrogen, chlorine, oxygen and sulphur may be written as :

