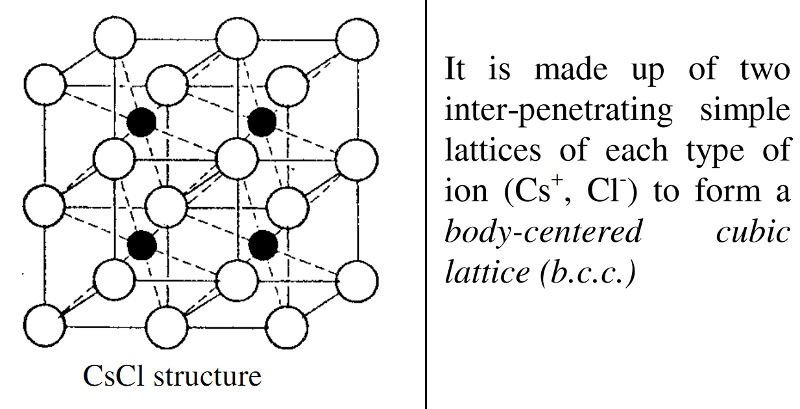
CsCl
In Caesium Chloride, CsCl, the radius ratio is 0.93.
This indicates a body-centred cubic type of arrangement, where each Cs+ ion is surrounded by eight Cl- ions, and vice versa.
The coordination is thus 8:8.
In a body-centred cubic arrangement, the atom at the centre of the cube is identical to those at the corners.
This structure is found in metals, but in CsCI if the ions at the corners are Cl- then there will be a Cs+ ion at the body-centred position.
The caesium chloride structure should be described as a body-centred cubic type of arrangement and not body-centred cubic.