Chemical Bonding
When two atoms are joined to make a chemical compound, the force of attraction between the two atoms is the chemical bond.
The way the atoms are held together (bonding) and their arrangement in space (structure) that accounts for distinct properties of different substances.
TYPES OF BONDS
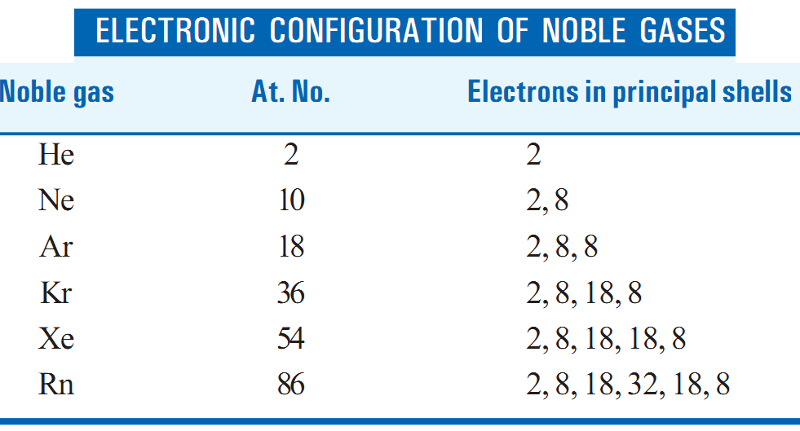
Atoms may attain a stable electronic configuration of noble gases in three different ways:
- losing electrons,
- gaining electrons,
- sharing electrons.
Elements may be divided into:
1. Electrpositive elements, whose atoms give up one or more electrons fairly readily.
2. Electronegative elements. which will accept electrons.
3. Elements which have little tendency to Jose or gain electrons.
Three different types of bond may be formed, depending on the electropositive or electronegative character of the atoms involved.

Ionic bonding involves the complete transfer of one or more electrons from one atom to another.
Covalent bonding involves the sharing of a pair of electrons between two atoms.
Metallic bonding the valency electrons are free to move throughout the whole crystal.
