Ionic bond
Ionic bonds are formed when electropositive elements react with electronegative elements.
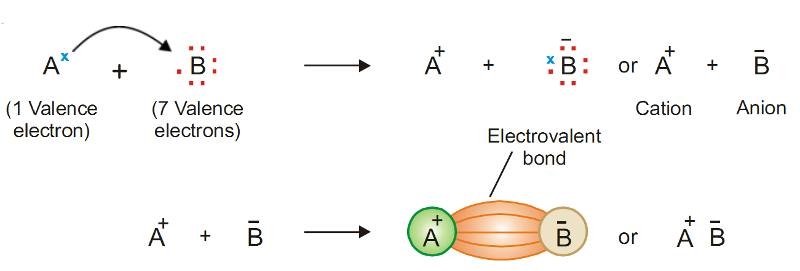
Two atoms are so unequal in their attraction for valence electrons that the more electronegative atom strips an electron completely away from its partner.
The two resulting oppositely charged atoms (or molecules) are called ions.
A positively charged ion is called a cation.
The negatively charged ion is called an anion.
Because of their opposite charges, cations and anions attract each other.
This attraction is called an ionic bond.
Ionic bonding, electrons are transferred before bond formation, forming an ion pair.
It typically involves a metal with a nonmetal
CONDITIONS FOR FORMATION OF IONIC BOND
The conditions favourable for the formation of an ionic bond are :
(1) Number of valence electrons
The atom A should possess 1, 2 or 3 valence electrons, while the atom B should have 5, 6 or 7 valence electrons.
The elements of group IA, IIA and IIIA satisfy this condition for atom A and those of groups VA, VIA, and VIIA satisfy this condition for atom B.
(2) Net lowering of Energy
To form a stable ionic compound, there must be a net lowering of the energy.
Energy must be released as a result of the electron transfer and formation of ionic compound by the
following steps :
(a) The removal of electron from atom A (A – e– → A+) requires input of energy, which is the ionization energy (IE).
It should be low.
(b) The addition of an electron to B (B + e– → B – ) releases energy, which is the electron affinity of B (EA). It should be high.
(c) The electrostatic attraction between A+ and B – in the solid compound releases energy, which is the electrical energy.
It should also be high.
If the energy released in steps (b) and (c) is greater than the energy consumed in step (a),
the overall process of electron transfer and formation of ionic compound results in a net release of energy.
Therefore, ionisation of A will occur and the ionic bond will be formed. For example, in case of formation of sodium chloride (NaCl), we have
The net energy released is 187 + 85 – 119 = 153 kcal. Since the overall process results in a lowering of energy, the ionic bond between Na and Cl will be formed.
(3) Electronegativity difference of A and B
From the line of argument used in (2), we can say that atoms A and B if they have greatly different electronegativities, only then they will form an ionic bond.
In fact, a difference of 2 or more is necessary for the formation of an ionic bond between atoms A and B.
Thus Na has electronegativity 0.9, while Cl has 3.0. Since the difference is (3.0 – 0.9) = 2.1, Na and Cl will form an ionic bond.
FACTORS GOVERNING THE FORMATION OF IONIC BOND
(1) Ionisation Energy
The ionisation energy of the metal atom which looses electron(s) should be low so that the formation of +vely charged ion is easier.
Lower the ionisation energy greater will be the tendency of the metal atom of change into cation and hence greater will be the ease of formation of ionic bond.
That is why alkali metals and alkaline earth metals form ionic bonds easily.
Out of these two, alkali metals form ionic bonds easily as compared to alkaline earth metals.
In a group the ionisation energy decreases as we move down the group and therefore, the tendency to form ionic bond increases in a group downward.
Due to this reason Cs is the most electropositive atom among the alkali metals.
(2) Electron Affinity
The atom which accepts the electron and changes into anion should have high electron affinity.
Higher the electron affinity more is the energy released and stable will be the anion formed.
The elements of group VI A and VII A have, in general, higher electron affinity and have high tendency to form ionic bonds.
Out of these two, the elements of group VII A (halogens) are more prone to the formation of ionic bond than the elements of group VI A.
In moving down a group the electron affinity decreases and, therefore, the tendency to form ionic bond also decreases.
(3) Lattice Energy
After the formation of cations and anions separately, they combine to form ionic compound.
In this process, energy is released. It is called Lattice Energy. It may be defined as "the amount of energy released when one mole of an ionic compound is formed from its cations and anions."
Greater the lattice energy, greater the strength of ionic bond.
The value of lattice energy depends upon the following two factors :
(a) Size of the ions
In order to have the greater force of attraction between the cations and anions their size should
be small as the force of attraction is inversely proportional to the square of the distance between them.
(b) Charge on Ions
Greater the charge on ions greater will be the force of attraction between them and, therefore,
greater will be the strength of the ionic bond.
Necessary for the formation of an ionic bond between atoms A and B. Thus Na has electronegativity
0.9, while Cl has 3.0. Since the difference is (3.0 – 0.9) = 2.1, Na and Cl will form an ionic bond.
