Shape or Structure of CH4 , NH3 and H2O- Tetrahedral geometry
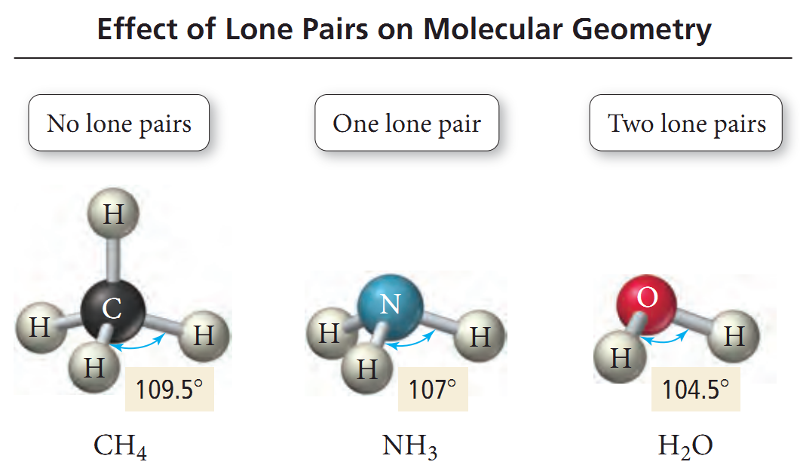
The ideal bond angle for Tetrahedral geometry is 109.5°.
If all four electron groups exerts equal repulsions on one another, the bond angles in the molecule would all be the ideal tetrahedral angle, 109.5°.
However, the actual angle between bonds is influenced by lone pair.
The lone pair occupies more of the angular space around a nucleus, exerting a greater repulsive force on neighboring electrons and compressing bond angles.
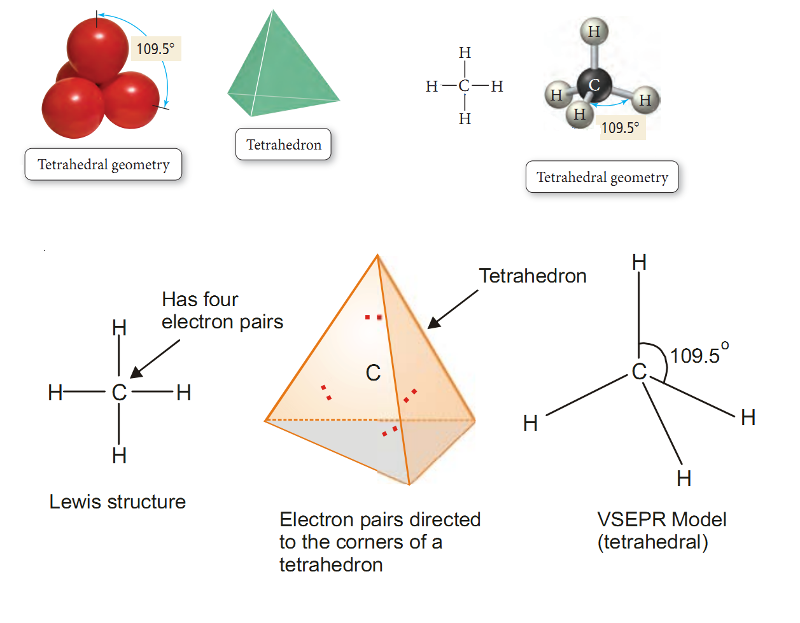
Methane, CH4.
Lewis structure of methane shows that the central C atom has four bonding electron pairs.
These electron pairs repel each other and are thus directed to the four corners of a regular tetrahedron.
A regular tetrahedron is a solid figure with four faces which are equilateral triangles.
All bond angles are 109.5º.
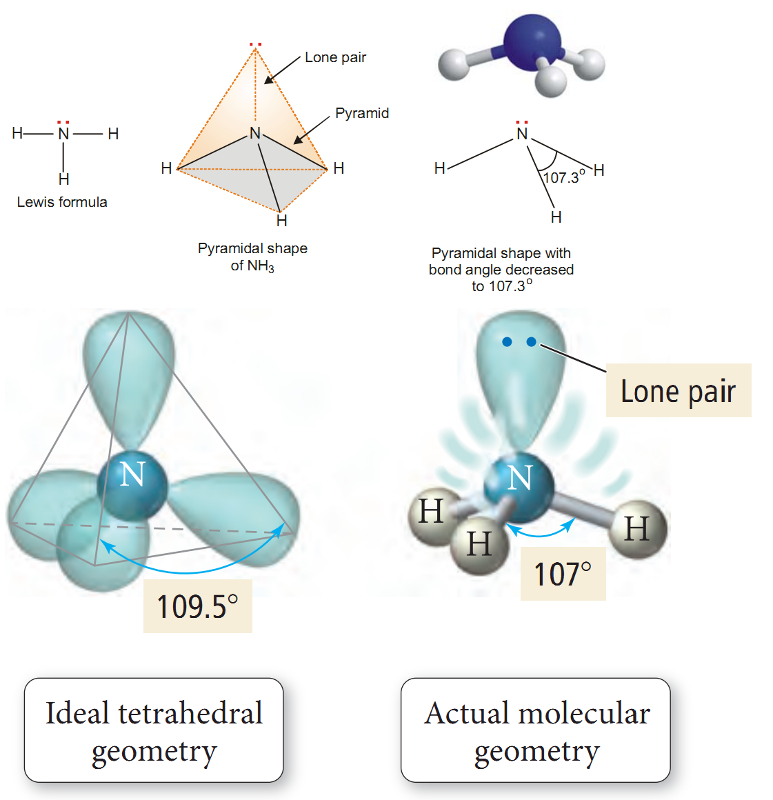
Ammonia molecule.
The Lewis structure of NH3 shows that the central N atom has three bonding electrons and one lone electron pair.
The VSEPR theory says that these electron pairs are directed to the corners of a tetrahedron.
Thus we predict that H–N–H bond angle should be 109.5º.
But the shape of a molecule is determined by the arrangement of atoms and not the unshared electrons.
Thus, if we see only at the atoms, we can visualise NH3 molecule as a pyramid with the N atom located at the apex and H atoms at the three corners of the triangular base.
According to VSEPR theory, a lone pair exerts greater repulsion on the bonding electron pairs than the bonding pairs do on each other.
As a result, the bonds of NH3 molecule are pushed slightly closer.
This explains why the observed bond angle H–N–H is found to be 107.3º instead of 109.5º predicted from tetrahedral geometry.
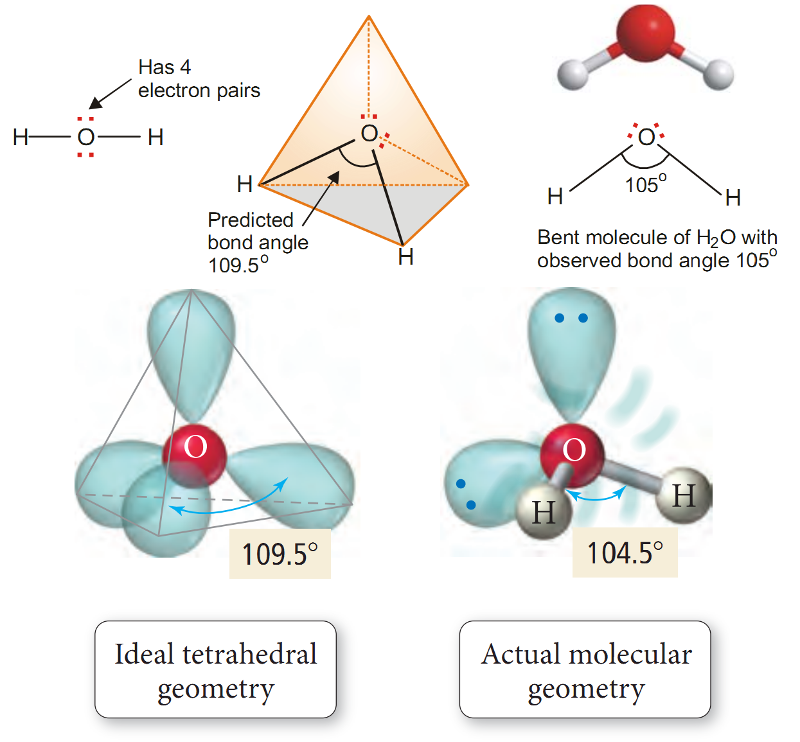
Water, H2O.
In the structural formula of H2O, the O atom is bonded to two H atoms by covalent bonds and has two lone pairs.
Thus O is surrounded by two bonding electron pairs and two unshared electron pairs.
VSEPR theory says that in order to secure maximum separation between them, the four electron pairs are directed to the corners of a tetrahedron.
If we look at the three atoms (and ignore the unshared pairs), the atoms HOH lie in the same plane and the predicted bond angle is 109.5º.
But with two unshared pairs repelling the bonding pairs, the bond angle is compressed to 105º, the experimental value.
Thus the H2O molecule is flat and bent at an angle at the O atom.
Such a molecule is called a bent molecule or angular molecule.