NaCl
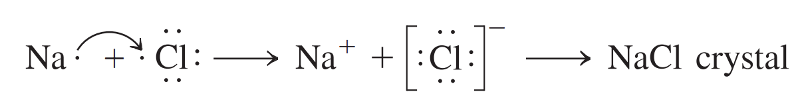
An atom of sodium (11Na) encounters an atom of chlorine (17Cl).
A sodium atom has a total of 11 electrons, with its single valence electron in the third electron shell.
A chlorine atom has a total of 17 electrons, with 7 electrons in its valence shell.
When these two atoms meet, the lone valence electron of sodium is transferred to the chlorine atom, and both atoms end up with their valence shells complete.
The sodium no longer has an electron in the third shell, the second shell is now the valence shell.
The electron transfer between the two atoms moves one unit of negative charge from sodium to chlorine.
Sodium, now with 11 protons but only 10 electrons, has a net electrical charge of 1+; the sodium atom has become a cation.
Conversely, the chlorine atom, having gained an extra electron, now has 17 protons and 18 electrons, giving it a net electrical charge of 1-; it has become a chloride ion—an anion.
Compounds formed by ionic bonds are called ionic compounds, or salts.
Nearly all ionic compounds are solids at normal temperatures and pressures.
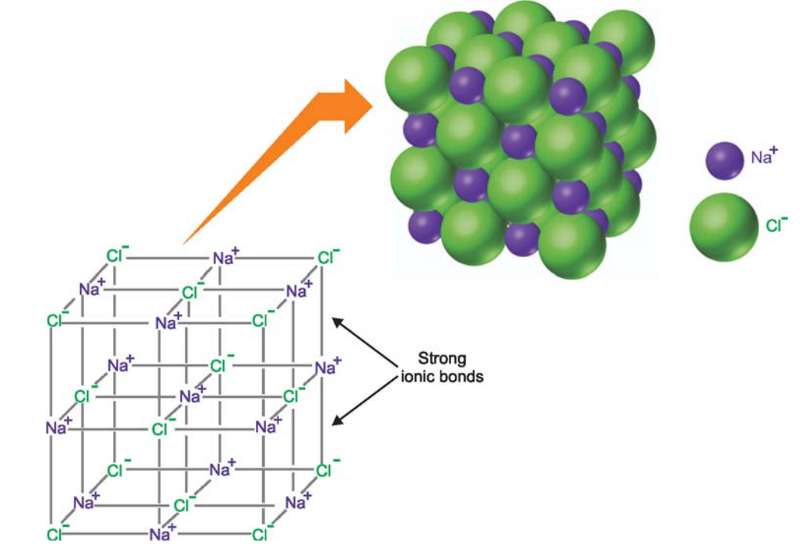
The solid has a definite geometric structure called a crystal.
Ions in a crystal are arranged so the potential energy resulting from the attractions and repulsions between them is at a minimum.
The precise form of the crystal depends on the kinds of ions in the compound, their sizes, and the ratio in which they appear.
The strong electrostatic forces (ionic bonds) hold the ions in fixed position in the crystal.
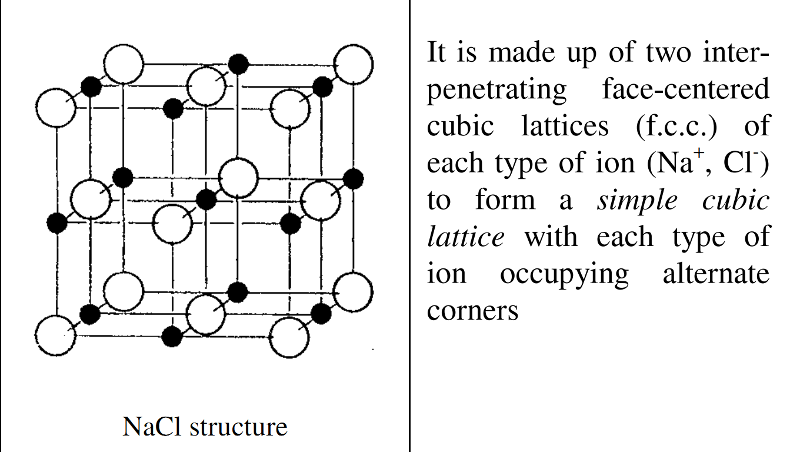
For sodium chloride, NaCl the radius ratio is 0.52.
It is an octahedral arrangement.
Each Na+ ion is surrounded by six Cl- ions at the corners of a regular octahedron and similarly each Cl- ion is surrounded by six Na+ ions.
The coordination is thus 6:6.
