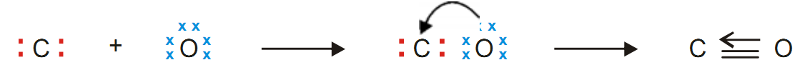Coordinate Bond
In a normal covalent bond, each of the two bonded atoms contributes one electron to make the shared pair.
In some cases, a covalent bond is formed when both the electrons are supplied entirely by one atom.
Such a bond is called co-ordinate covalent or dative bond.
It may be defined as :
a covalent bond in which both electrons of the shared pair come from one of the two atoms (or ions).
The compounds containing a coordinate bond are called coordinate compounds.
If an atom A has an unshared pair of electrons (lone pair) and another atom B is short of two electrons than the stable number, coordinate bond is formed.
A donates the lone pair to B which accepts it.
Thus both A and B achieve the stable 2 or 8 electrons, the lone pair being held in common.
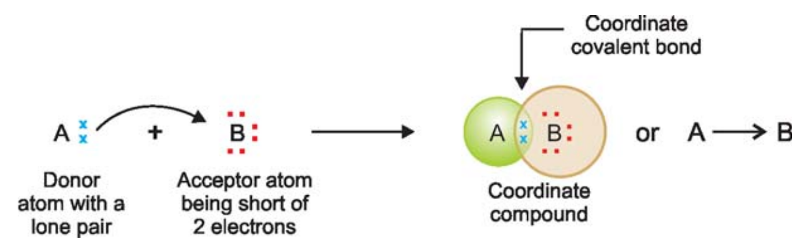
The atom A which donates the lone pair is called the donor, while B which accepts it the acceptor.
The bond thus established is indicated by an arrow pointing from A to B. Although the arrow head indicates the origin of the electrons.
Once the coordinate bond is formed it is in no way different from an ordinary covalent bond.
EXAMPLES OF COORDINATE COMPOUNDS OR IONS
Ammonium ion, NH4+
In ammonia molecule, the central N atom is linked to three H atoms and yet N has an unshared pair of electrons.
The H+ ion furnished by an acid has no electron to contribute and can accept a pair of electrons loaned by N atom.
Thus, NH3 donates its unshared electrons to H+ forming ammonium ion.
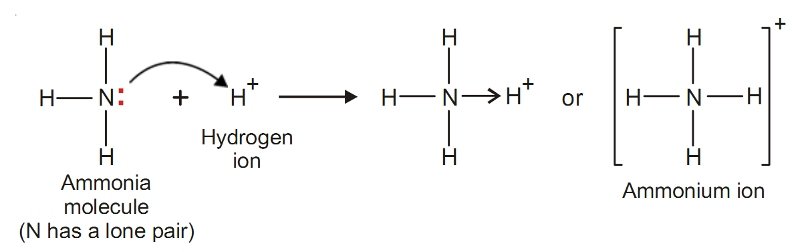
All the N–H bonds in NH4+are identical, once the coordinate bond N→H+ is established.
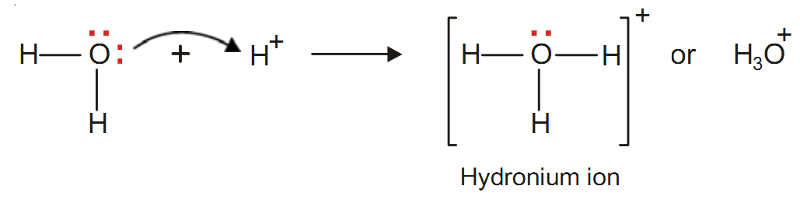
Hydronium ion, H3O+
The oxygen atom in water molecule is attached to two H atoms by two covalent bonds.
There are still two unshared pairs of electrons with the O atom.
The O atom donates one of these pairs of electrons to H+ ion and the hydronium ion is thus formed.
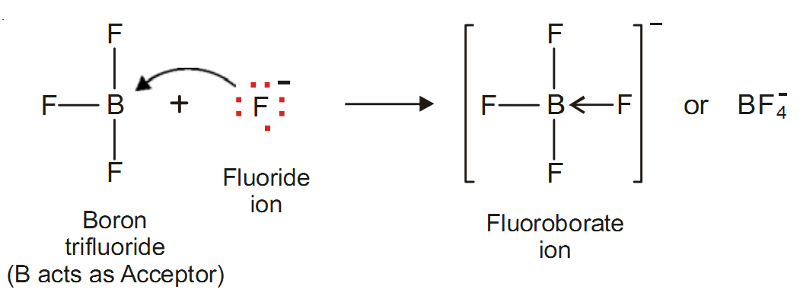
Fluoroborate ion, BF4+
It is formed when a boron trifluoride molecule (BF3) shares a pair of electrons supplied by fluoride ion (F–).
Carbon Monoxide, CO
Carbon atom has four valence electrons while oxygen atom has six.
By forming two covalent bonds between them, O atom achieves octet but C atom has only six electrons.
Therefore O donates an unshared pair of electron to C, and a coordinate covalent bond is established between the two atoms.
Lewis structure of CO may be written as