Non Covalent Bonds - Van Der Waal's forces
A covalent bond in which electrons are shared unequally and the bonded atoms acquire a partial positive and negative charge, is called a polar covalent bond.
This results in weak intermolecular attraction forces that are termed as non-covalent bonds.
The examples are Van der Waal's forces and Hydrogen bonding.
Van Der Waal forces and Dipoles
A molecule having partial positive and negative charge separated by a distance is commonly referred to as a Dipole (two poles).
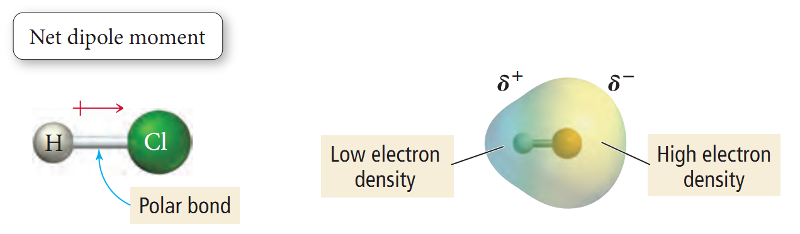
The dipole of a bond is indicated by an arrow from positive to negative end with a crossed tail as shown above in HCl molecule.
Since two atoms of different elements do not have exactly the same attraction for electrons in a bond, all bonds between unlike atoms are polar to some extent.
The amount of polarity of a bond is determined by the difference of electronegativity (or tendency to attract electrons) of the two bonded atoms.
The greater the difference of electronegativity between two atoms, greater the polarity.
The Van Der Waal interaction term includes:
1. force between permanent dipoles (Keesom force) - Dipole:Dipole interaction
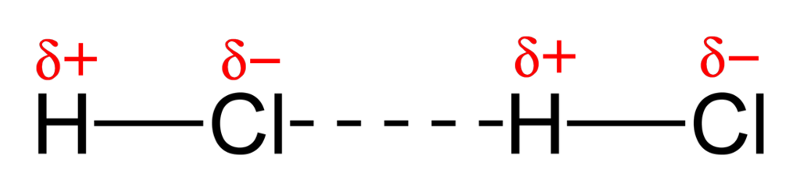
2. force between a permanent dipole and a corresponding induced dipole (Debye force) - Dipole:Induced Dipole Interaction

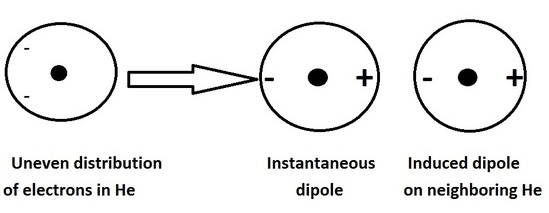
3. force between instantaneously induced dipoles (London dispersion force) - Induced Dipole:Induced Dipole interaction.
The dipole of a bond is indicated by an arrow from positive to negative end with a crossed tail as shown above in HCl molecule.
Since two atoms of different elements do not have exactly the same attraction for electrons in a bond, all bonds between unlike atoms are polar to some extent.
The amount of polarity of a bond is determined by the difference of electronegativity (or tendency to attract electrons) of the two bonded atoms.
The greater the difference of electronegativity between two atoms, greater the polarity.
