Chemical Bonding : Molecular Shapes of CH4, NH3, H2O, BeCl2 and BF3
The SIDGWICK-POWELL THEORY preceding the VSEPR model explained 3D geometry of the molecule.
SIDGWICK-POWELL THEORY
The approximate shape can be predicted from the number of electron pairs in the outer or valence shell of the central atom.
The outer shell contains one or more bond pairs of electrons, but it may also contain unshared pairs of electrons (lone pairs).
Bond pairs and Ione pairs were taken as equivalent, since all electron pairs take up some space, and since all electron pairs repel each other.
Repulsion is minimized if the electron pairs are orientated in space as far apart as possible.
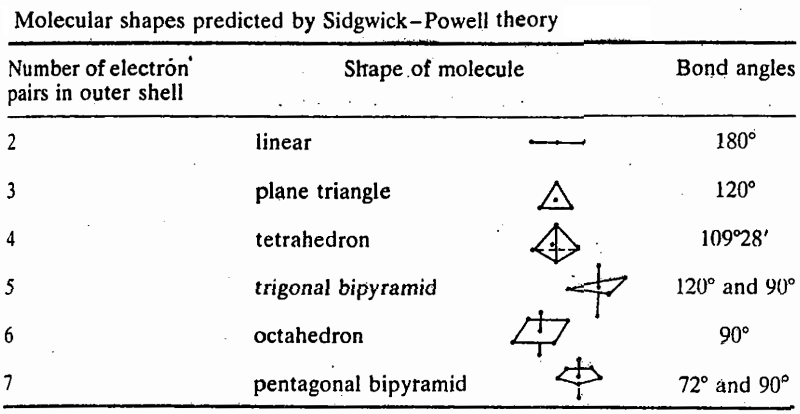
1. If there are two pairs of electrons in the valence shell of the central atom, the orbitals containing them will be oriented at 180° to each other. It follows that if these orbitals overlap with orbitals from other atoms to form bonds, then the molecule formed will be linear.
2. If there are three electron pairs on the central atom, they will be at 120° to each other, giving a plane triangular structure.
3. For four electron pairs the angle is 109"28', and the shape is tetrahedral.
4. For five pairs, the shape is a trigonal bipyramid.
5. For six pairs the angles are 90° and the shape is octahedral.
