Covalent Bond
Langmuir (1919) refined the Lewis postulations by abandoning the idea of the stationary cubical arrangement of the octet, and by introducing the term covalent bond.
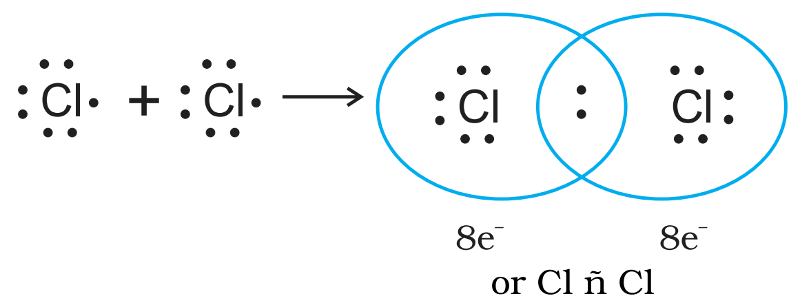
The Lewis-Langmuir theory can be understood by considering the formation of the chlorine molecule,Cl2.
The Cl atom with electronic configuration, [Ne]3s2 3p5, is one electron short of the argon configuration.
The formation of the Cl2 molecule can be understood in terms of the sharing of a pair of electrons between the two chlorine atoms. Each chlorine atom contributing one electron to the shared pair.
In the process both chlorine atoms attain the outer shell octet of the nearest noble gas (i.e., argon).
The Lewis dot structures can be written for other molecules also, in which the combining atoms may be identical or different.
The important conditions being that:
• Each bond is formed as a result of sharing of an electron pair between the atoms.
• Each combining atom contributes at least one electron to the shared pair.
• The combining atoms attain the outer-shell noble gas configurations as a result of the sharing of electrons.
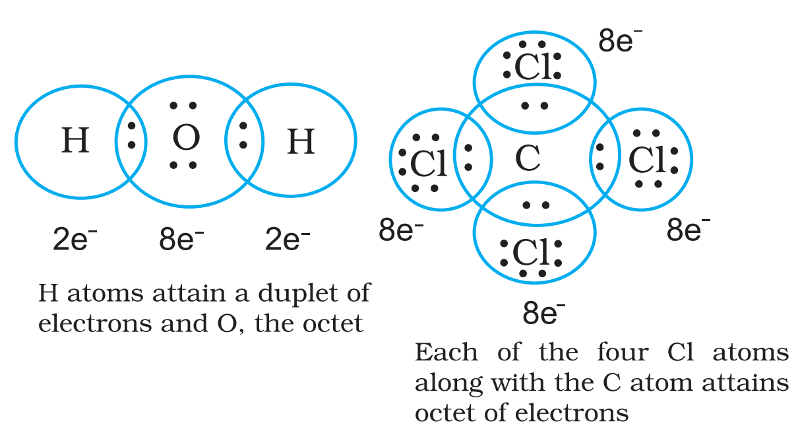
• Thus in water and carbon tetrachloride molecules, formation of covalent bonds can be represented as:
CONDITIONS FOR FORMATION OF COVALENT BOND
The conditions favourable for the formation of covalent bonds are :
(1) Number of valence electrons
Each of the atoms A and B should have 5, 6 or 7 valence electrons so that both achieve the stable octet by sharing 3, 2 or 1 electron-pair.
Hydrogen has one electron in the valence shell and attains duplet.
The non-metals of groups VA, VIA and VIIA respectively satisfy this condition.
(2) Equal electronegativity
The atom A will not transfer electrons to B if both have equal electronegativity, and hence electron sharing will take place.
This can be strictly possible only if both the atoms are of the same element.
(3) Equal sharing of electrons
The atoms A and B should have equal (or nearly equal) electron affinity so that they attract the bonding electron pair equally.
Thus equal sharing of electrons will form a nonpolar covalent bond.
Of course, precisely equal sharing of electrons will not ordinarily occur except when atoms A and B are atoms of the same element, for no two elements have exactly the same electron affinity.
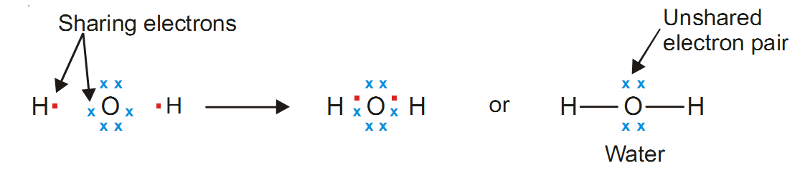
Example of Covalent Bond - Water, H2O
Oxygen atom (2, 6) has six valence electrons and can achieve the stable octet by sharing two electrons, one with each H atom.
Thus Lewis structure of water can be written as :
Example of Covalent Bond - Ammonia, NH3
Nitrogen atom (2, 5) has five valence electrons and can achieve the octet by sharing three electrons, one each with three H atoms.
This gives the following Lewis structure for ammonia :
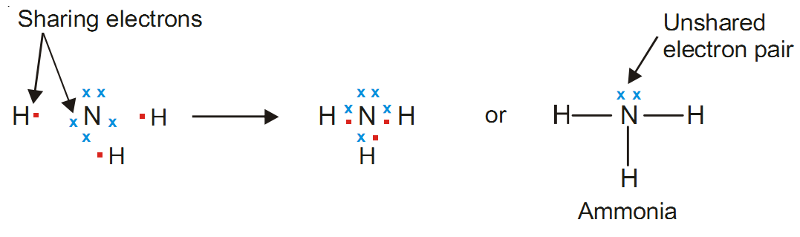
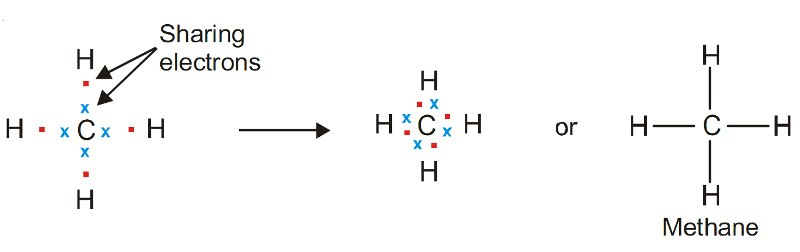
Example of Covalent Bond - Methane, CH4
Carbon atom (2, 4) has four electrons in the valence shell. It can achieve the stable octet by sharing these electrons with four H atoms, one with each H atom.
Thus the Lewis structure of methane can be written as :